|
If you have any other queries of CBSE Class 11 Chemistry Structure of Atom MCQs Multiple Choice Questions with Answers, feel free to reach us so that we can revert back to us at the earliest possible. Hope the information shed above regarding NCERT MCQ Questions for Class 11 Chemistry Chapter 2 Structure of Atom with Answers Pdf free download has been useful to an extent. The charge on an electron was found by oil drop exp. When an electron jumps from 4th orbit to 2nd orbit, it gives rise toġ0. The energy of each photon is directly proportional to the frequency.ĩ. An atom is like a watermelon of positive charge in which electrons are embedded like the seeds.Ĩ. The angular momentum of a moving electron in an orbit is quantised.ħ. Position and momentum of a small particle cannot be measured simultaneously with absolute accuracy.Ħ. An orbital can have maximum two electrons.ĥ. Orbitals are filled in order of increasing energy.Ĥ. The order of occupation is as follows: 1s<2s<2p<3s<3p<4s<3d<4p<5s<4d<5p<6s<4f<5d<6p<7s<5f<6d<7p Another way to view this order of increasing energy is by using Madelung's Rule: Figure 1. Degenrate orbitals are first singly occupied.ģ. The Aufbau Principle, also called the building-up principle, states that electron's occupy orbitals in order of increasing energy. The maximum number of electrons which can be accommodated in an orbital is 2n². 
Two electrons can have the same values for n, l and m but should have different values of s. 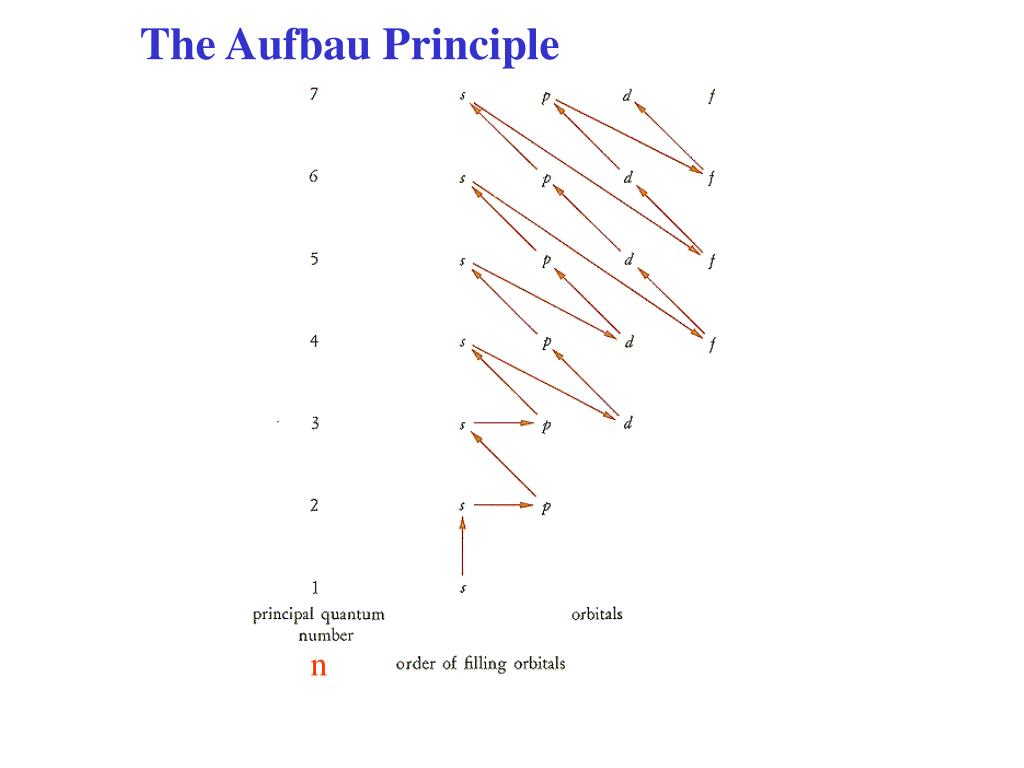
Answerīohr’s atom consists of circular orbits. The angular momentum of a moving electron is an integral multiple of h/2π. 
Answerįor s-orbitals, orbital angular momentum is zero. AnswerĪngular momentum of electron in a shell is quantized, was derived by Bohr. The energy of an electron in 3d orbital is less than that in the 4s orbital in the hydrogen atom. AnswerĢnd orbital is larger in size than Is orbital. Answerįor a given value of l, the shape of orbital is independent of the value of n. Three electrons in p-orbital remains unpaired. The energy of the electron in 3d orbital is less than that in 4s orbital. The nucleus of an atom contains electrons and protons. (d) \(\frac \) is the angular momentum of the electron in the ………………. How many moles of electrons weigh one kilogram? AnswerĪnswer: (d) helium nuclei which impinged on a metal foil and got scattered. (d) helium nuclei which impinged on a metal foil and got scattered. (c) helium atoms which impinged on a metal foil and got reflected (b) γ-rays, which impinged on a metal foil and got absorbed (a) β-particles which impinged on a metal foil and got absorbed Rutherford’s experiment which established the nuclear model of the atom used a beam of Structure of Atom Class 11 MCQs Questions with Answers Refer to the Structure of Atom Class 11 MCQs Questions with Answers here along with a detailed explanation. Practice MCQ Questions for Class 11 Chemistry with Answers on a daily basis and score well in exams. The maximum number of electrons in any shell is 2n2, where n is the principal quantum number.We have compiled the NCERT MCQ Questions for Class 11 Chemistry Chapter 2 Structure of Atom with Answers Pdf free download covering the entire syllabus. 
Passing from one element to another of the next higher atomic number, one proton and one electron are added each time to the neutral atom. If double occupation does occur, the Pauli exclusion principle requires that electrons that occupy the same orbital must have different spins (+1⁄2 and −1⁄2). Neon (Z 10) asked in Quantum Mechanical Model of Atom by Rajan01 (46. Hund's rule asserts that if multiple orbitals of the same energy are available, electrons will occupy different orbitals singly and with the same spin before any are occupied doubly. Using Aufbau principle, write the ground state electronic configuration of following atoms. An example is the configuration 1s2 2s2 2p6 3s2 3p3 for the phosphorus atom, meaning that the 1s subshell has 2 electrons, and so on.Įlectron behavior is elaborated by other principles of atomic physics, such as Hund's rule and the Pauli exclusion principle. In this way, the electrons of an atom or ion form the most stable electron configuration possible. For example, the 1s subshell is filled before the 2s subshell is occupied. The aufbau principle (, from the German Aufbauprinzip, which means "building-up principle"), also called the aufbau rule, states that in the ground state of an atom or ion, electrons fill subshells of the lowest available energy, then they fill subshells of higher energy. Wikipedia (0.00 / 0 votes) Rate this definition:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
